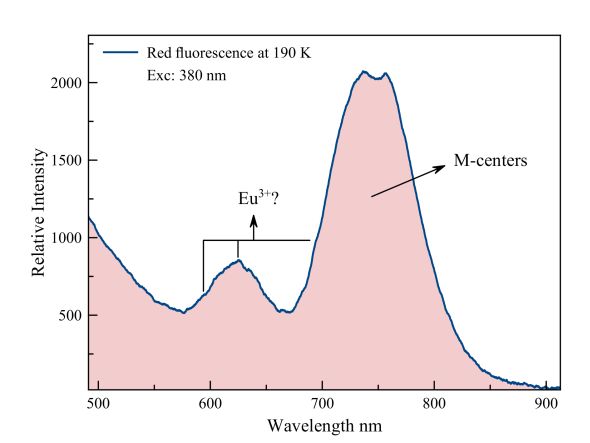
The dark red fluorescence of the Seilles fluorite has two important peaks. A broad peak is found on the border of red and infrared, between 700 nm and 840 nm. This is the fluorescence of the M-centers. M-centers consist of two F-centers (Farbenzentrum). These are vacancies in the Crystal where actually a halogen, fluorine-ion should be in this case. These blank spots are surrounded by positive ions and can therefore easily capture an electron. For adjoining F-centers, charge compensation often happens by a metal ion or a proton. The total of two F-centers with a metal ion is an M-center. At 700 nm, we see a shoulder on the emission of the M-centers and also at 595 nm and 622 nm two peaks are united. Gorobets assigns these to trivalent europium.
Measured with: Ocean Optics USB4000 spectrometer and a LED light source of 380 nm
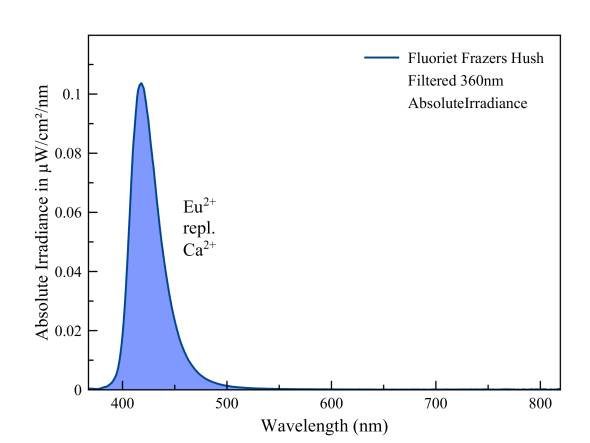
The strong blue fluorescence of fluorite is caused by divalent europium that partially replaces calcium.
You may download the raw data of this measurement by clicking on the icon.
Fluorite Seilles, red fluorescence
Fluorite Weardale, blue fluorescence
Measured with: Ocean Optics Flame spectrometer, a reflection probe and a LED light source of 360 nm
This spectrum has been split up for the sake of detail. The peaks were attributed to the emissions of the many rare earth elements (lanthanoids) present. Attribution of the peaks to the fluorophores was done using the tables that were compiled by Boris S. Gorobets and Alexandre A. Rogojine in 'Luminescent Spectra of Minerals - Reference Book' (Moscow 2002). The attributions are probably correct but should be considered as approximative and possibly incomplete rather than absolute.
Fluorite, var.: yttrofluorite, Hundholmen, Tysfjord, Nordland, Norway
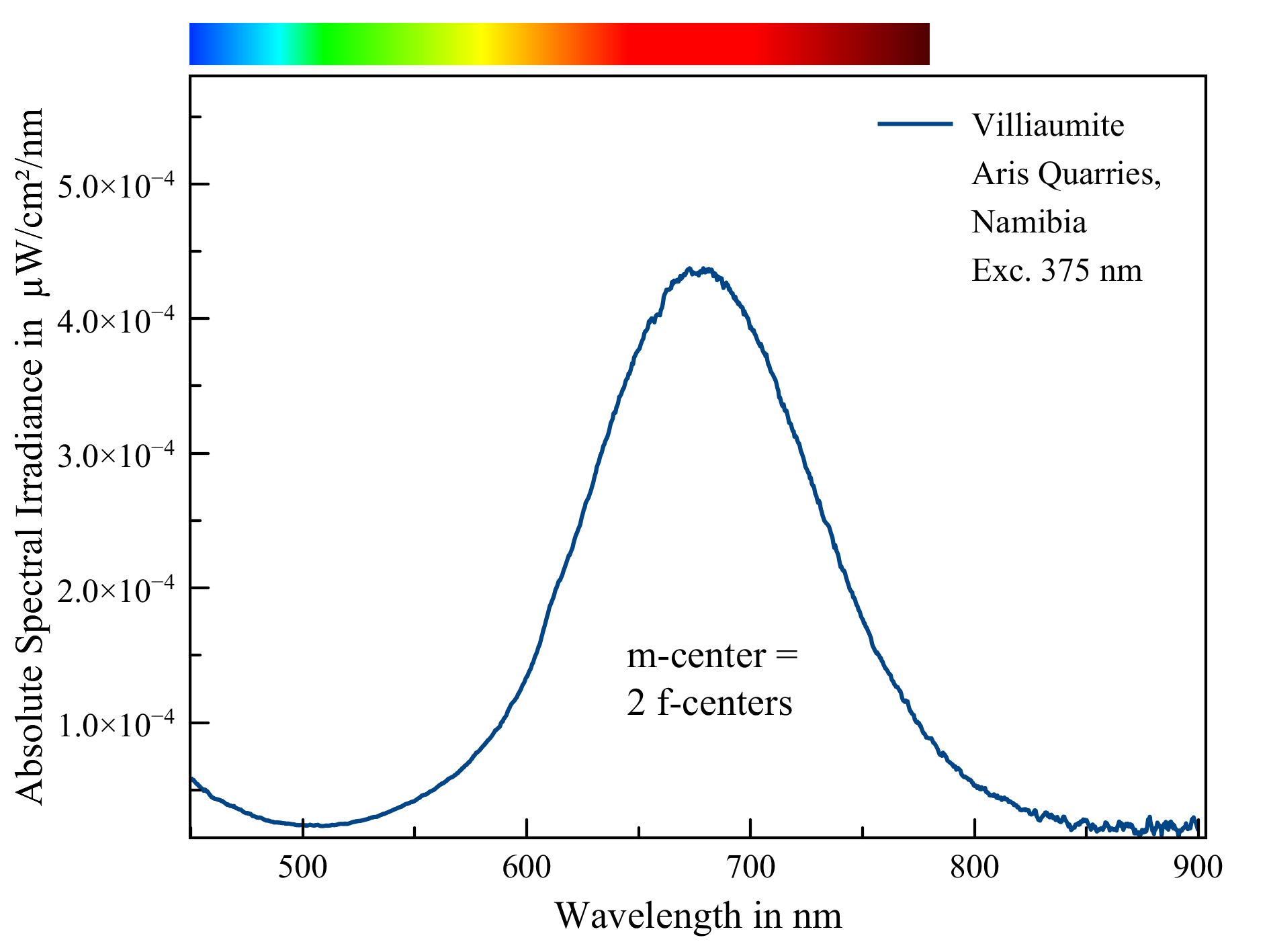
Villiaumite, Aris Quarries, Namibia
The fluorescence of villiaumite is caused by m-centers. These m-centers consist of a pair of f-centers (farbzenter). An f-center, in its turn, is a vacancy in the crystal lattice where an anion is missing and an electron is occupying the void.